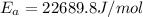Chemistry, 22.02.2021 18:40 danbat3023
The rate constant for a certain reaction is measured at two different temperatures:
temperature k
376.0 °C 4.8 x 10^8
280.0 °C 2.3 x 10^8
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy Ea for this reaction.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3
You know the right answer?
The rate constant for a certain reaction is measured at two different temperatures:
temperature k
Questions

Mathematics, 29.12.2020 22:50

Mathematics, 29.12.2020 22:50


Mathematics, 29.12.2020 22:50



Advanced Placement (AP), 29.12.2020 23:00

Computers and Technology, 29.12.2020 23:00

Mathematics, 29.12.2020 23:00



English, 29.12.2020 23:00


Mathematics, 29.12.2020 23:00

Mathematics, 29.12.2020 23:00




Chemistry, 29.12.2020 23:00

Geography, 29.12.2020 23:00

![ln \frac{k_{2}}{k_{1}} = \frac{-E_{a}}{R}[\frac{1}{T_{2}} - \frac{1}{T_{1}}]](/tpl/images/1136/3217/c502b.png)
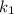 = rate constant at temperature
= rate constant at temperature 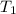 =
= 
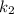 = rate constant at temperature
= rate constant at temperature 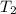 =
= 
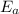 = activation energy = ?
= activation energy = ?
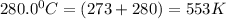
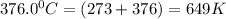
![ln \frac{4.8\times 10^8}{2.3\times 10^8} = \frac{-E_{a}}{8.314}[\frac{1}{649} - \frac{1}{553}]](/tpl/images/1136/3217/fcab8.png)