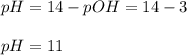Chemistry, 19.02.2021 01:00 urstruulyemily
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamine is . Calculate the pH of the base solution after the chemist has added of the solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added. g

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamin...
Questions

Biology, 26.08.2019 14:00



Health, 26.08.2019 14:00

Biology, 26.08.2019 14:00

Mathematics, 26.08.2019 14:00

Mathematics, 26.08.2019 14:00








Health, 26.08.2019 14:00

English, 26.08.2019 14:00

Biology, 26.08.2019 14:00


Geography, 26.08.2019 14:00

Mathematics, 26.08.2019 14:00

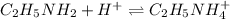
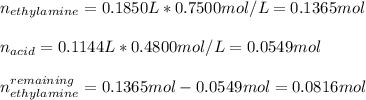
![[ethylamine]=\frac{0.0816mol}{0.1850L+0.1144L}=0.2725M](/tpl/images/1129/1639/9c401.png)
![[salt]=\frac{0.0549mol}{0.1850L+0.1144L}=0.1834M](/tpl/images/1129/1639/c810c.png)
![pOH=pKb+log(\frac{[salt]}{[base]} )\\\\pOH=3.19+log(\frac{0.1834M}{0.2725M})\\\\pOH=3.0](/tpl/images/1129/1639/77c7e.png)