
Chemistry, 18.02.2021 09:20 kaylamount
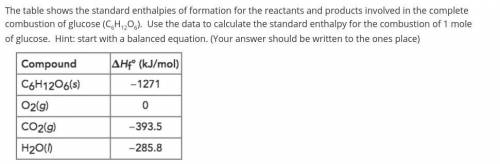
The table shows the standard enthalpies of formation for the reactants and products involved in the complete combustion of glucose (C6H12O6). Use the data to calculate the standard enthalpy for the combustion of 1 mole of glucose. Hint: start with a balanced equation. (Your answer should be written to the ones place)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 05:20
Why does the sun appear to be the brightest star in the sky? a- its apparent brightness is much greater than other stars. b- it burns more gas, making it brighter than any other star. c- it is the largest star in the galaxy, so it is the brightest star. d- its relative distance to earth is closer than the other stars.
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3
You know the right answer?
The table shows the standard enthalpies of formation for the reactants and products involved in the...
Questions

Advanced Placement (AP), 30.06.2019 23:30



Mathematics, 30.06.2019 23:30

Mathematics, 30.06.2019 23:30

Advanced Placement (AP), 30.06.2019 23:30




History, 30.06.2019 23:30

Biology, 30.06.2019 23:30




Spanish, 30.06.2019 23:30



English, 30.06.2019 23:30

History, 30.06.2019 23:30

History, 30.06.2019 23:30



