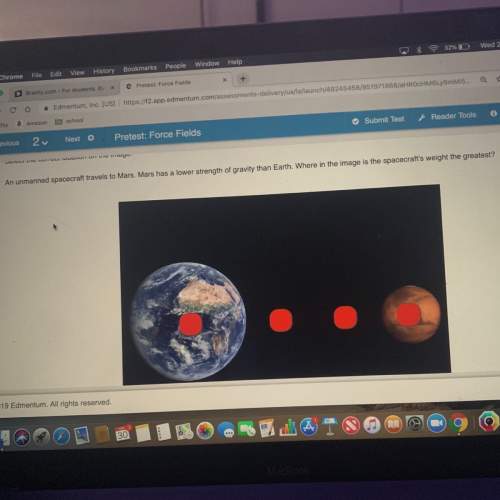
Chemistry, 18.02.2021 01:00 anthonywdjr5211
A 3.81-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO3 → Na2CO3 + H2CO3
In this experiment, carbon dioxide and water vapors combine to form H2CO3. After decomposition, the Na2CO3 had a mass of 2.86 grams.
Determine the mass of the H2CO3 produced.
Calculate the percentage yield of H2CO3 for the reaction. Show your work or describe the calculation process in detail.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
A 3.81-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO3 → Na2CO3 + H2CO3
Questions


English, 03.12.2019 03:31








Mathematics, 03.12.2019 03:31

Biology, 03.12.2019 03:31


Mathematics, 03.12.2019 03:31


Computers and Technology, 03.12.2019 03:31

Mathematics, 03.12.2019 03:31

Chemistry, 03.12.2019 03:31

English, 03.12.2019 03:31

Mathematics, 03.12.2019 03:31