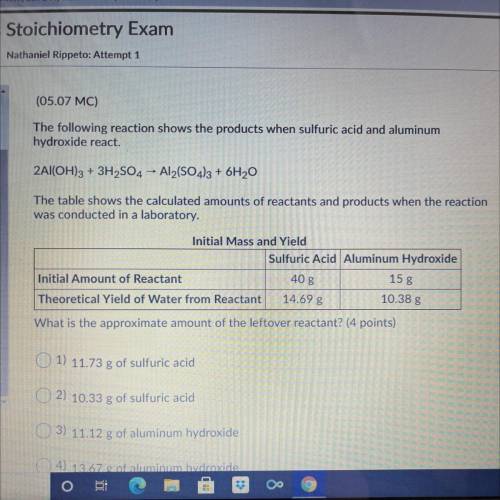
The following reaction shows the products when sulfuric acid and aluminum
hydroxide react.
2A...

The following reaction shows the products when sulfuric acid and aluminum
hydroxide react.
2Al(OH)3 + 3H2SO4 - Al2(SO4)3 + 6H20
The table shows the calculated amounts of reactants and products when the reaction
was conducted in a laboratory.
What is the approximate amount of the leftover reactant?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1
You know the right answer?
Questions

Mathematics, 22.03.2020 05:53

Mathematics, 22.03.2020 05:53



Mathematics, 22.03.2020 05:54

Mathematics, 22.03.2020 05:54

Mathematics, 22.03.2020 05:54



Mathematics, 22.03.2020 05:56


Mathematics, 22.03.2020 05:56


Mathematics, 22.03.2020 05:57



Chemistry, 22.03.2020 05:57

Mathematics, 22.03.2020 05:58

Mathematics, 22.03.2020 05:58



