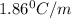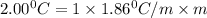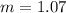Chemistry, 17.02.2021 09:20 milkshakegrande101
What is the molal concentration of a sucrose (C12H22011) solution whose freezing point is -2.00 °C? The Kf for water is 1.86 °C/m.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:10
Describe the difference between a. a hypothesis and a theory and b. an observation and an experiment.
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1
You know the right answer?
What is the molal concentration of a sucrose (C12H22011) solution whose freezing point is
-2.00 °C?...
Questions



English, 26.05.2021 19:00

History, 26.05.2021 19:00

Chemistry, 26.05.2021 19:00


Mathematics, 26.05.2021 19:00

Mathematics, 26.05.2021 19:00

World Languages, 26.05.2021 19:00

History, 26.05.2021 19:00



Business, 26.05.2021 19:00





English, 26.05.2021 19:00

English, 26.05.2021 19:00

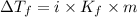
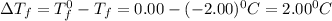 = Depression in freezing point
= Depression in freezing point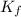 = freezing point constant =
= freezing point constant =