
Chemistry, 17.02.2021 01:10 gui00g7888888888888
For the reaction below, the order of reaction for NH4+ is 1, and the order of reaction for NO2 is 1.
NH4(aq) + NO3(aq) = N2(g) + 2H2O(1)
What is the rate law for the reaction?
A. R=k[NH+4] [NO-2]
B. R= [NH+4] [NO-2]2
C. R=k[NH]4 [NO]2
D. R=K[N2][H20]
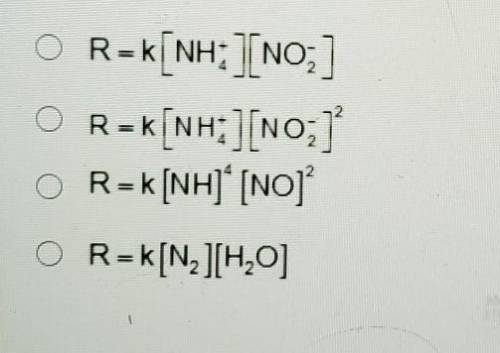

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is lincoln's purpose in writing this speech? question 1 options: to stress the difficulties of war to honor those who died in the war to call for an end to the war to call the country to join a new war
Answers: 1

Chemistry, 21.06.2019 22:30
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3


Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2
You know the right answer?
For the reaction below, the order of reaction for NH4+ is 1, and the order of reaction for NO2 is 1....
Questions

Mathematics, 22.07.2019 06:00





Mathematics, 22.07.2019 06:00



Mathematics, 22.07.2019 06:00

Mathematics, 22.07.2019 06:00

Social Studies, 22.07.2019 06:00



Mathematics, 22.07.2019 06:00

Mathematics, 22.07.2019 06:00




Physics, 22.07.2019 06:00




