
Chemistry, 16.02.2021 18:50 screamqueen
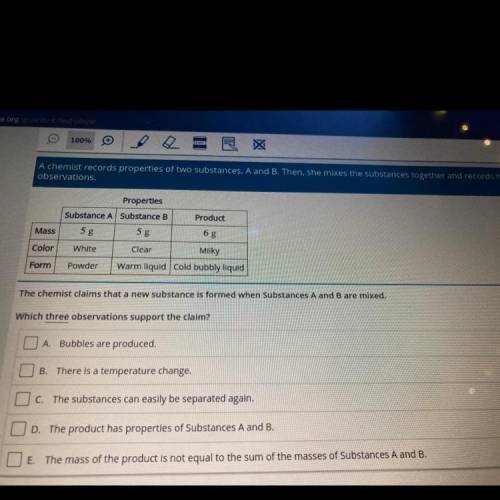
A chemist records properties of two substances, A and B. Then, she mixes the substances together and records her
observations.
Product
Properties
Substance A Substance B
5 g
5 g
White
Clear
Mass
Color
Milky
Form
Powder
Warm liquid cold bubbly liquid
The chemist claims that a new substance is formed when Substances A and B are mixed.
Which three observations support the claim?
O A Bubbles are produced.
B
There is a temperature change.
O c. The substances can easily be separated again.
OD. The product has properties of Substances A and B.
O E. The mass of the product is not equal to the sum of the masses of Substances A and B.

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

You know the right answer?
A chemist records properties of two substances, A and B. Then, she mixes the substances together and...
Questions


Mathematics, 31.01.2020 01:45




History, 31.01.2020 01:45

History, 31.01.2020 01:45

Biology, 31.01.2020 01:45


Computers and Technology, 31.01.2020 01:45


Mathematics, 31.01.2020 01:45

Spanish, 31.01.2020 01:45



Mathematics, 31.01.2020 01:45


History, 31.01.2020 01:45




