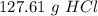Chemistry, 15.02.2021 04:10 gulleraliyeva1999
one reaction that produces hydrogen gas can be represented by the unbalanced chemical equation Mg(s)+HCI(aq) -> MgCI(aq)+H2(g). What is the mass of HCI is consumed by the reaction of 3.25 mol of magnesium?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2

Chemistry, 23.06.2019 01:30
Ascientist is measuring the pressure that is exerted by each of the following gases in the atmosphere: carbon dioxide, oxygen, and nitrogen. which term most likely describes what she is measuring?
Answers: 1
You know the right answer?
one reaction that produces hydrogen gas can be represented by the unbalanced chemical equation Mg(s)...
Questions



English, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Business, 12.12.2020 17:00

Health, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Chemistry, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00

History, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Chemistry, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

History, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Computers and Technology, 12.12.2020 17:00

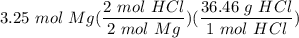 [S - DA] Multiply/Divide [Cancel out units]:
[S - DA] Multiply/Divide [Cancel out units]: