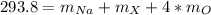Chemistry, 13.02.2021 07:50 naomicervero
1.25 pts An ionic compound has the formula Na XO4, where X represents an unknown transition metal. The compound has a molar mass of approximately 293.8 g/mol. Which of the following elements is the most likely identity of X?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Find the empirical formula of each of the following compounds. given mass or for each element in a sample of the compound 3,611 g ca; 6.389 g c1
Answers: 1

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 05:10
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3
You know the right answer?
1.25 pts
An ionic compound has the formula Na XO4, where X represents an unknown transition metal....
Questions

Mathematics, 26.10.2019 20:43

Chemistry, 26.10.2019 20:43




Mathematics, 26.10.2019 20:43




English, 26.10.2019 20:43


Chemistry, 26.10.2019 20:43

Biology, 26.10.2019 20:43

Biology, 26.10.2019 20:43




Biology, 26.10.2019 20:43

Mathematics, 26.10.2019 20:43

English, 26.10.2019 20:43