Zoom in
1 An astronaut who specializes in chemistry
discovers a strange chemical compound our...

Chemistry, 13.02.2021 05:10 JAYDENJONES0111
Zoom in
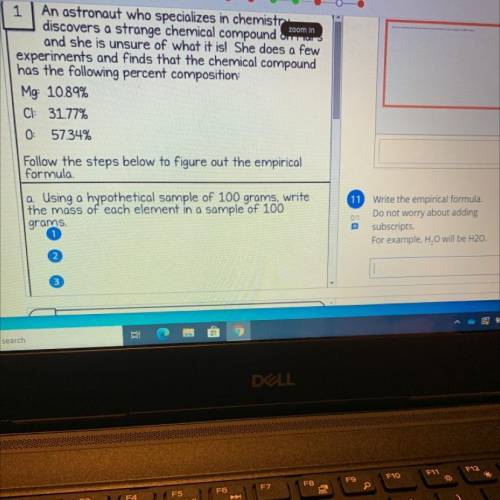
1 An astronaut who specializes in chemistry
discovers a strange chemical compound our
and she is unsure of what it is! She does a few
experiments and finds that the chemical compound
has the following percent composition:
Mg: 10.89%
CI: 31.77%
0 57.34%
Follow the steps below to figure out the empirical
formula
a Using a hypothetical sample of 100 grams, write
the mass of each element in a sample of 100
grams
11 Write the empirical formula.
01
Do not worry about adding
subscripts.
For example, H. O will be H20.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3
You know the right answer?
Questions


Mathematics, 05.02.2020 04:54


Mathematics, 05.02.2020 04:54

Mathematics, 05.02.2020 04:54



Biology, 05.02.2020 04:54

History, 05.02.2020 04:54

Mathematics, 05.02.2020 04:54

Biology, 05.02.2020 04:54

Biology, 05.02.2020 04:54






Mathematics, 05.02.2020 04:54

Mathematics, 05.02.2020 04:54




