
Chemistry, 12.02.2021 05:00 blakesmith0110
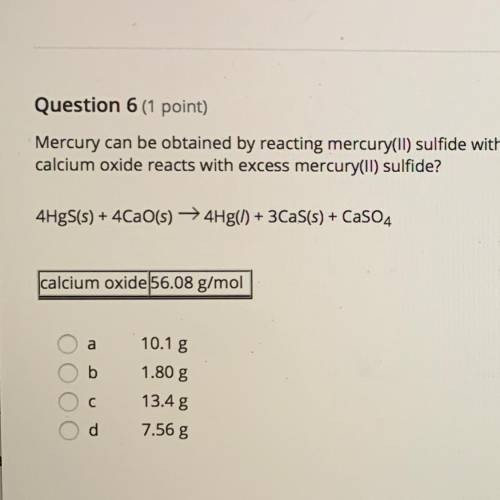
Mercury can be obtained by reacting mercury(II) sulfide with calcium oxide. How many grams of Mercury metal are produced when 2.11 g of calcium oxide reacts with excess mercury(II) sulfide?
4HgS+4CaO=4Hf+3CaS+CaSO4
a. 10.1 g
b. 1.80 g
c. 13.4 g
d. 7.56 g

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

You know the right answer?
Mercury can be obtained by reacting mercury(II) sulfide with calcium oxide. How many grams of Mercur...
Questions

Physics, 01.12.2021 19:40

Social Studies, 01.12.2021 19:40

Mathematics, 01.12.2021 19:40

Mathematics, 01.12.2021 19:40


Social Studies, 01.12.2021 19:40



Computers and Technology, 01.12.2021 19:40


History, 01.12.2021 19:40




History, 01.12.2021 19:40

History, 01.12.2021 19:40

Chemistry, 01.12.2021 19:40



Business, 01.12.2021 19:40



