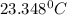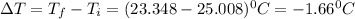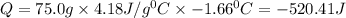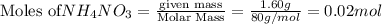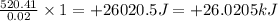Chemistry, 12.02.2021 01:40 pgfrkypory2107
In a coffee cup calorimeter, 1.60 g of NH4NO3 is mixed with 75.0 g of water at an initial temperature of 25.008C. After dissolution of the salt, the final temperature of the calorimeter contents is 23.348C. Assuming the solution has a heat capacity of 4.18 J 8C21 g21 and assuming no heat loss to the calorimeter, calculate the enthalpy change for the dissolution of NH4NO3 in units of kJ/mol.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
You know the right answer?
In a coffee cup calorimeter, 1.60 g of NH4NO3 is mixed with 75.0 g of water at an initial temperatur...
Questions

English, 21.10.2020 05:01


Mathematics, 21.10.2020 05:01

Mathematics, 21.10.2020 05:01

Mathematics, 21.10.2020 05:01

English, 21.10.2020 05:01

Mathematics, 21.10.2020 05:01

History, 21.10.2020 05:01

Mathematics, 21.10.2020 05:01


English, 21.10.2020 05:01




Mathematics, 21.10.2020 05:01



Mathematics, 21.10.2020 05:01

Arts, 21.10.2020 05:01

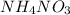 is +26.0205 kJ/mol
is +26.0205 kJ/mol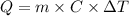
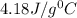
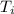 =
= 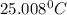
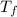 =
=