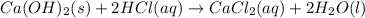Chemistry, 11.02.2021 23:20 babygirl123468
A sample of 7.34 g of solid calcium hydroxide is added to 34.5 mL of 0.380 M Aqueous hydrochloric acid. Write the balanced chemical equation for the reaction. Physical states are optional.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2


Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1
You know the right answer?
A sample of 7.34 g of solid calcium hydroxide is added to 34.5 mL of 0.380 M Aqueous hydrochloric ac...
Questions

English, 14.12.2021 02:50



Mathematics, 14.12.2021 02:50


Biology, 14.12.2021 02:50



Biology, 14.12.2021 02:50


Arts, 14.12.2021 02:50

Chemistry, 14.12.2021 02:50

SAT, 14.12.2021 02:50

Social Studies, 14.12.2021 02:50



Mathematics, 14.12.2021 02:50

Business, 14.12.2021 02:50

SAT, 14.12.2021 02:50