
Chemistry, 10.02.2021 22:20 gracethegreat1
Identify both of the PE wells in the graph below as belonging to either a covalent bond or an LDF. Then, explain what this means regarding the energy needed to break a covalent bond.
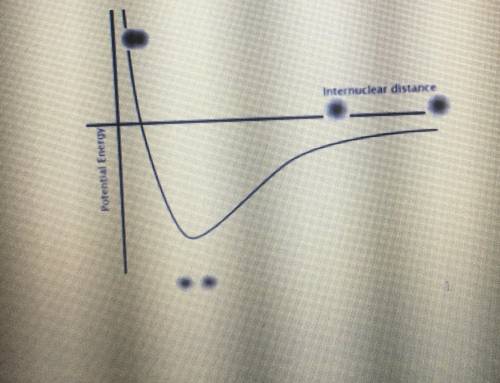

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2
You know the right answer?
Identify both of the PE wells in the graph below as belonging to either a covalent bond or an LDF. T...
Questions

Advanced Placement (AP), 02.08.2019 01:30


Health, 02.08.2019 01:30


Health, 02.08.2019 01:30

Mathematics, 02.08.2019 01:30


History, 02.08.2019 01:30

Mathematics, 02.08.2019 01:30





History, 02.08.2019 01:30

Mathematics, 02.08.2019 01:30


History, 02.08.2019 01:30






