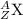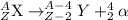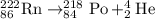Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
You know the right answer?
The nuclear equation below represents the alpha decay of 222/86 Rn --> 4/2 He + X
What is the ma...
Questions

Mathematics, 04.12.2019 02:31


Mathematics, 04.12.2019 02:31



History, 04.12.2019 02:31

Mathematics, 04.12.2019 02:31

Biology, 04.12.2019 02:31

History, 04.12.2019 02:31



Health, 04.12.2019 02:31

Mathematics, 04.12.2019 02:31



History, 04.12.2019 02:31

Mathematics, 04.12.2019 02:31