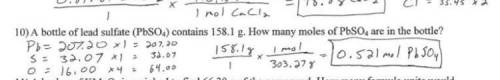Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
A bottle contains 158.1 grams of lead (II) sulfate (PbSO4), used for car batteries. How many moles d...
Questions

Mathematics, 24.03.2021 16:50

World Languages, 24.03.2021 16:50


Social Studies, 24.03.2021 16:50

Mathematics, 24.03.2021 16:50






Mathematics, 24.03.2021 16:50

Arts, 24.03.2021 16:50


Mathematics, 24.03.2021 16:50

Physics, 24.03.2021 16:50




Mathematics, 24.03.2021 16:50