
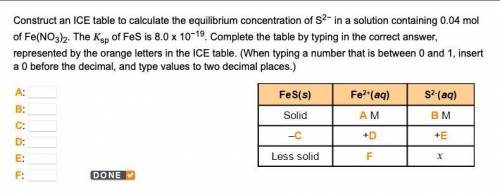
Construct an ICE table to calculate the equilibrium concentration of S2− in a solution containing 0.04 mol of Fe(NO3)2. The Ksp of FeS is 8.0 x 10−19. Complete the table by typing in the correct answer, represented by the orange letters in the ICE table. (When typing a number that is between 0 and 1, insert a 0 before the decimal, and type values to two decimal places.)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3


Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1
You know the right answer?
Construct an ICE table to calculate the equilibrium concentration of S2− in a solution containing 0....
Questions

Mathematics, 18.04.2021 19:00

English, 18.04.2021 19:00

Mathematics, 18.04.2021 19:00

Chemistry, 18.04.2021 19:00





English, 18.04.2021 19:10

Mathematics, 18.04.2021 19:10

Mathematics, 18.04.2021 19:10


Mathematics, 18.04.2021 19:10



Mathematics, 18.04.2021 19:10

Mathematics, 18.04.2021 19:10



Mathematics, 18.04.2021 19:10



