
Chemistry, 08.02.2021 19:00 mervindavisk
Both diamond and graphite (i. e. pencil lead) consist of carbon atoms. They are only different in their crystalline structures. One carat or 0.20 g, of a high-quality diamond costs up to $5000, while 25 g of pencil lead may only cost $2. Determine the numbers of C-atom in diamond vs. graphite which can be obtained with $1.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3
You know the right answer?
Both diamond and graphite (i. e. pencil lead) consist of carbon atoms. They are only different in th...
Questions


Mathematics, 21.03.2020 03:15




English, 21.03.2020 03:15



Mathematics, 21.03.2020 03:15




Biology, 21.03.2020 03:15

Mathematics, 21.03.2020 03:15


Mathematics, 21.03.2020 03:16

Mathematics, 21.03.2020 03:16


Mathematics, 21.03.2020 03:16

Mathematics, 21.03.2020 03:16

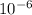 mol
mol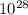 atom
atom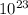 Atoms
Atoms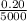
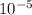 g Carbon
g Carbon

