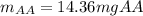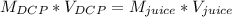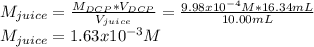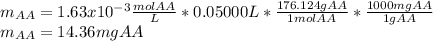Chemistry, 08.10.2019 18:30 annadel742
Students performed a procedure similar to part ii of this experiment (analyzing juices for vitamin c content) as described in the procedure section. three 10.00ml samples of juice were titrated with dcp that had a standardized concentration of 9.98x10-4m. the three titrations took an average of 16.34ml of dcp. calculate the mass (in mg) in 50.00ml of juice. (mm ascorbic acid = 176.124 g/mol)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2


Chemistry, 22.06.2019 13:50
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3
You know the right answer?
Students performed a procedure similar to part ii of this experiment (analyzing juices for vitamin c...
Questions

English, 02.12.2020 23:30



Computers and Technology, 02.12.2020 23:30





Mathematics, 02.12.2020 23:30



Mathematics, 02.12.2020 23:30


Chemistry, 02.12.2020 23:30

Mathematics, 02.12.2020 23:30


Mathematics, 02.12.2020 23:30

Physics, 02.12.2020 23:30

Mathematics, 02.12.2020 23:30

Physics, 02.12.2020 23:30