Chemistry, 07.02.2021 08:20 skyvargasov9cad
What is the theoretical yield of SO3 produced by 8.96 g of S?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2


Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2
You know the right answer?
What is the theoretical yield of SO3 produced by 8.96 g of S?...
Questions




History, 02.08.2019 19:30








Health, 02.08.2019 19:30




Biology, 02.08.2019 19:30



Biology, 02.08.2019 19:30

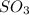 produced by 8.96 g of S is 33.6 g
produced by 8.96 g of S is 33.6 g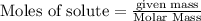
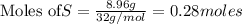
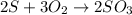
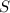 produce = 3 moles of
produce = 3 moles of 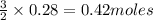 of
of