
Chemistry, 07.02.2021 08:10 linsey9201
How many grams of ZrCl4 can be produced if 123 g of ZrSiO4 react with 85.0 g of Cl2?
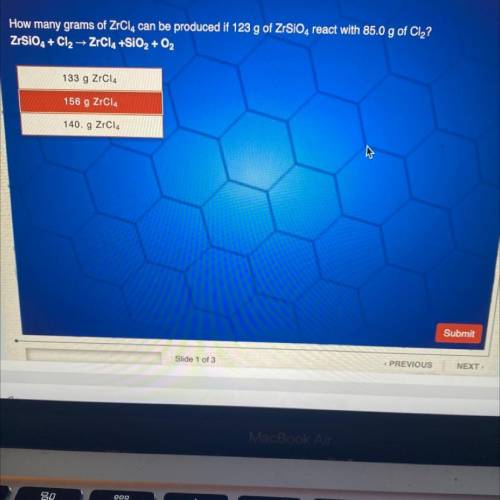

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1
You know the right answer?
How many grams of ZrCl4 can be produced if 123 g of ZrSiO4 react with 85.0 g of Cl2?
...
...
Questions





Mathematics, 17.09.2019 11:20





Social Studies, 17.09.2019 11:20

Mathematics, 17.09.2019 11:20

Health, 17.09.2019 11:20


History, 17.09.2019 11:20



Social Studies, 17.09.2019 11:20

Geography, 17.09.2019 11:30





