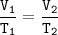Chemistry, 06.02.2021 07:50 jaimejohnston2
An arctic weather balloon is filled with 4.96 L of helium gas inside a prep shed. The temperature inside the shed is 6. °C. The balloon is then taken outside, where the temperature is 2. °C. Calculate the new volume of the balloon.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
An arctic weather balloon is filled with 4.96 L of helium gas inside a prep shed. The temperature in...
Questions

Social Studies, 04.08.2019 06:30

Biology, 04.08.2019 06:30

History, 04.08.2019 06:30

Biology, 04.08.2019 06:30

English, 04.08.2019 06:30

History, 04.08.2019 06:30


History, 04.08.2019 06:30






Spanish, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


SAT, 04.08.2019 06:30



Spanish, 04.08.2019 06:30