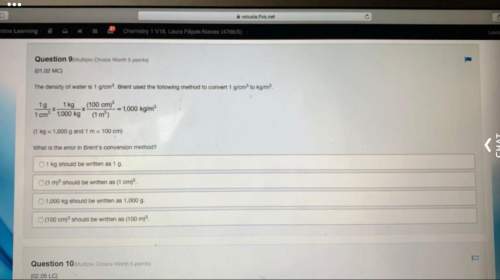
Chemistry, 06.02.2021 04:30 korireidkdotdot82021
a reaction vessel contains a small piece of magnesium metal and air (20%) of which is oxygen gas. The mixture is ignited and burns with a burst of light and heat, producing solid magneisum oxide. The mass of mgO, however, is less than the initital mass of magnesium and oxygen combined. What loss of mass

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1


You know the right answer?
a reaction vessel contains a small piece of magnesium metal and air (20%) of which is oxygen gas. Th...
Questions




History, 13.01.2021 14:00



Advanced Placement (AP), 13.01.2021 14:00

Mathematics, 13.01.2021 14:00


Mathematics, 13.01.2021 14:00

Mathematics, 13.01.2021 14:00




Mathematics, 13.01.2021 14:00