
Chemistry, 05.02.2021 22:00 catsareokiguess
A solid sample of Zinc Hydroxide is added to 0.350 L of 0.500 M aqueous Hydrogen Bromide. The solution that remains is still acidic. It is then titrated with 0.500 M NaOH solution, and it takes 88.5 mL of the NaOH solution to reach the equivalence point. What mass of Zinc Hydroxide was added to the Hydrogen Bromide solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1


Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

You know the right answer?
A solid sample of Zinc Hydroxide is added to 0.350 L of 0.500 M aqueous Hydrogen Bromide. The soluti...
Questions






Mathematics, 30.07.2019 23:30




History, 30.07.2019 23:30

Business, 30.07.2019 23:30




Mathematics, 30.07.2019 23:30


English, 30.07.2019 23:30

English, 30.07.2019 23:30


English, 30.07.2019 23:30

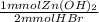 = 65.375 mmol Zn(OH)₂.
= 65.375 mmol Zn(OH)₂.

