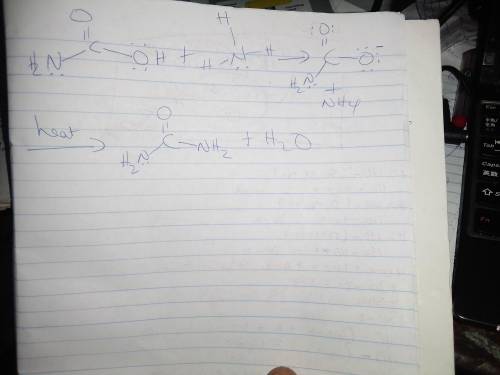Chemistry, 05.02.2021 22:00 saucyboyFredo
Here is the reaction of carbamic acid and ammonia to form an amide and water. There is a scheme of a reversible reaction where carbamic acid reacts with ammonia to give water and an unknown compound. Carbamic acid is H2NCOH with an oxygen atom attached to the carbon atom by a double bond. Ammonia is a nitrogen atom with three H atoms attached. Water is HOH. Draw the amide product of this reaction.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

You know the right answer?
Here is the reaction of carbamic acid and ammonia to form an amide and water. There is a scheme of a...
Questions

History, 23.10.2019 07:00




Mathematics, 23.10.2019 07:00



Biology, 23.10.2019 07:00

Mathematics, 23.10.2019 07:00


Chemistry, 23.10.2019 07:00

Mathematics, 23.10.2019 07:00


Mathematics, 23.10.2019 07:00

History, 23.10.2019 07:00

Mathematics, 23.10.2019 07:00

Physics, 23.10.2019 07:00

Arts, 23.10.2019 07:00