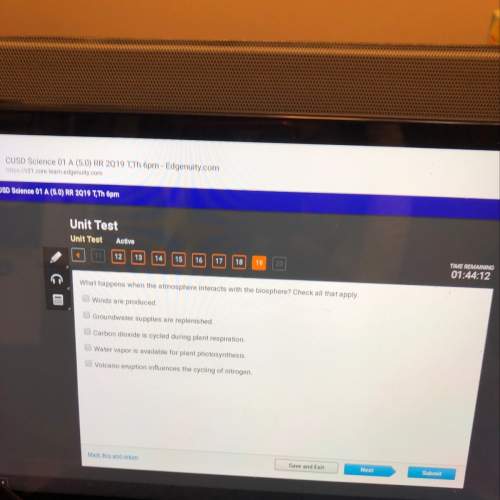
Suppose of nickel(II) bromide is dissolved in of a aqueous solution of potassium carbonate. Calculate the final molarity of nickel(II) cation in the solution. You can assume the volume of the solution doesn't change when the nickel(II) bromide is dissolved in it. Round your answer to significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

You know the right answer?
Suppose of nickel(II) bromide is dissolved in of a aqueous solution of potassium carbonate. Calculat...
Questions

Social Studies, 28.01.2022 15:10

Mathematics, 28.01.2022 15:10


Chemistry, 28.01.2022 15:10

Advanced Placement (AP), 28.01.2022 15:20




Social Studies, 28.01.2022 15:20

Geography, 28.01.2022 15:20

Mathematics, 28.01.2022 15:20

Engineering, 28.01.2022 15:20

Spanish, 28.01.2022 15:20


Mathematics, 28.01.2022 15:20


History, 28.01.2022 15:20

History, 28.01.2022 15:20


English, 28.01.2022 15:20