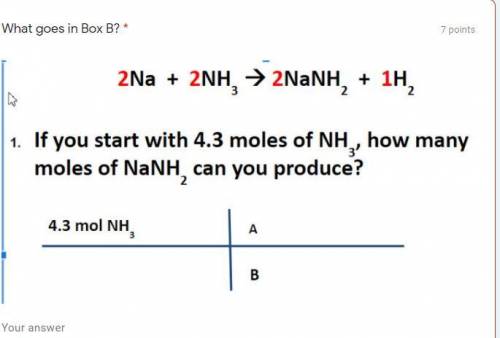
If you start with 4.3 moles of NH3, how many moles of NANH2 can be produced
...


Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

You know the right answer?
Questions







Mathematics, 05.09.2021 22:50




Biology, 05.09.2021 22:50


Mathematics, 05.09.2021 22:50


Spanish, 05.09.2021 22:50


Geography, 05.09.2021 23:00

Arts, 05.09.2021 23:00