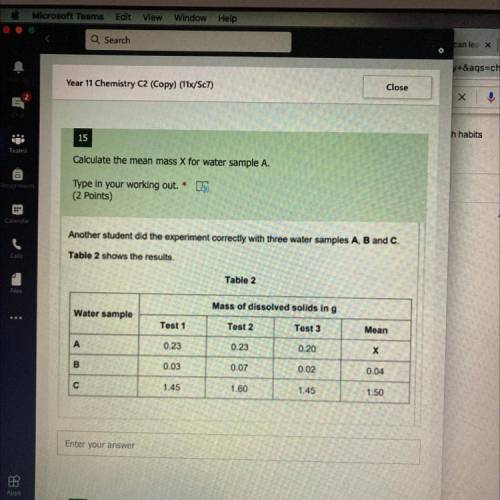
Answer pls pls plss this is my mock
...

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3


Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
Questions

Mathematics, 07.02.2022 23:20


Biology, 07.02.2022 23:20

Advanced Placement (AP), 07.02.2022 23:20



Biology, 07.02.2022 23:20


Mathematics, 07.02.2022 23:20

Mathematics, 07.02.2022 23:20

Mathematics, 07.02.2022 23:20

Mathematics, 07.02.2022 23:20






History, 07.02.2022 23:30