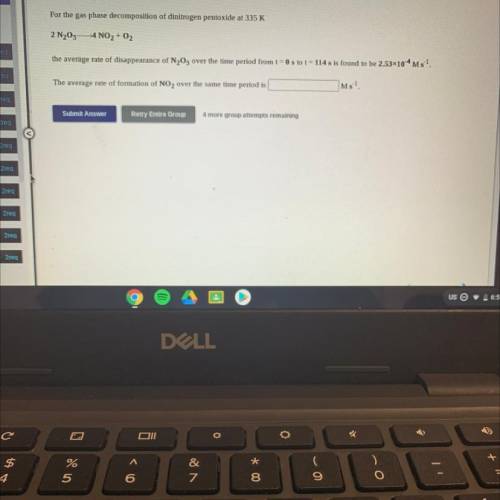
For the gas phase decomposition of dinitrogen pentoxide at 335 K
2 N205– 4 NO2 + O2
the avera...

For the gas phase decomposition of dinitrogen pentoxide at 335 K
2 N205– 4 NO2 + O2
the average rate of disappearance of N205 over the time period from t = 0 s to t = 114 s is found to be 2.53x10-4 M 5-1.
The average rate of formation of NO2 over the same time period is
Ms-1
Submit Answer
Retry Entire Group
4 more group attempts remaining

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

You know the right answer?
Questions

Mathematics, 14.10.2019 17:40

Chemistry, 14.10.2019 17:40

English, 14.10.2019 17:40

Mathematics, 14.10.2019 17:40

History, 14.10.2019 17:40



Geography, 14.10.2019 17:40

History, 14.10.2019 17:40


Social Studies, 14.10.2019 17:40

Mathematics, 14.10.2019 17:40


Mathematics, 14.10.2019 17:40



History, 14.10.2019 17:40


Mathematics, 14.10.2019 17:40

Mathematics, 14.10.2019 17:40



