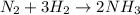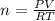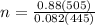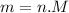Suppose a chemical engineer studying a new catalyst for the Haber reaction finds that liters per second of dinitrogen are consumed when the reaction is run at and the dinitrogen is supplied at . Calculate the rate at which ammonia is being produced. Give your answer in kilograms per second. Be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3


You know the right answer?
Suppose a chemical engineer studying a new catalyst for the Haber reaction finds that liters per sec...
Questions

Health, 12.07.2019 20:30


Social Studies, 12.07.2019 20:30

Mathematics, 12.07.2019 20:30


Mathematics, 12.07.2019 20:30


History, 12.07.2019 20:30

Mathematics, 12.07.2019 20:30


Mathematics, 12.07.2019 20:30


Mathematics, 12.07.2019 20:30

Mathematics, 12.07.2019 20:30


Business, 12.07.2019 20:30



Mathematics, 12.07.2019 20:30