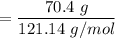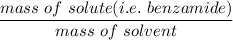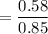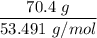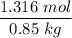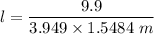Chemistry, 01.02.2021 20:40 alyonaprotopopova
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 2.7 C lower than the freezing point of pure X. On the other hand, when 70.4 g of ammonium chloride (NH CI) are dissolved in the same mass of X, the freezing point of the solution is 9.9 °C lower than the freezing point of pure X.
Required:
Calculate the van't Hoff factor for ammonium chloride in X.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3


Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezin...
Questions

Mathematics, 28.11.2019 19:31


Mathematics, 28.11.2019 19:31