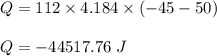What is the total amount of heat released in kilojoules when 112.0 g water at 50.0∘C cools to form ice at −45.0∘C? Use the following values for calculations, as needed.
Properties of Water−−−−−−−−−−−−−−−−−−Specific Heats(∘C)gas=1.84 J/g∘Cliquid=4.184 J/g∘Csolid=2.09 J/g∘C
Heat of VaporizationΔHvap=40.7 kJ/molHeat of FusionΔHfus=6.01 kJ/mol

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2
You know the right answer?
What is the total amount of heat released in kilojoules when 112.0 g water at 50.0∘C cools to form i...
Questions

Biology, 23.11.2019 07:31

Chemistry, 23.11.2019 07:31


History, 23.11.2019 07:31


Mathematics, 23.11.2019 07:31








Mathematics, 23.11.2019 07:31





Mathematics, 23.11.2019 07:31

English, 23.11.2019 07:31