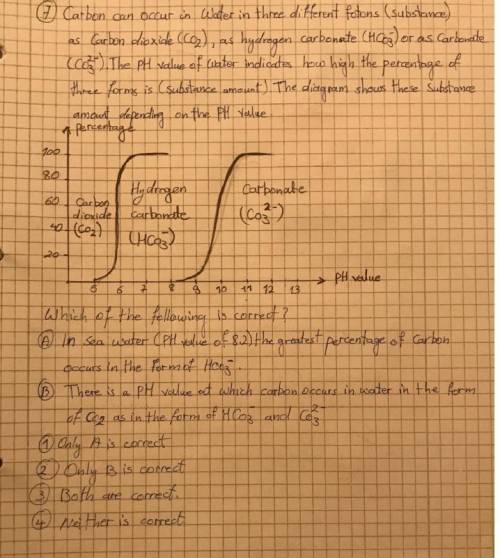
Can you help me please?
Thank you
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 20:10
Insoluble sulfide compounds are generally black in color. which of the following combinations could yield a black precipitate? check all that apply. na2s(aq)+kcl(aq) li2s(aq)+pb(no3)2(aq) pb(clo3)2(aq)+nano3(aq) agno3(aq)+kcl(aq) k2s(aq)+sn(no3)4(aq)
Answers: 1

Chemistry, 23.06.2019 13:30
How does water evaporating from a glass show that matter is made up of particles? a. the heat energy from the air causes the glass to fill up with water particles. b. the liquid water particles turn into water vapor that spreads in the air. c. the particles of the glass dissolve in water and cause it to evaporate. d. the tiny particles of the glass evaporate and seem to disappear.
Answers: 2
You know the right answer?
Questions



Social Studies, 11.03.2021 18:00

Mathematics, 11.03.2021 18:00

History, 11.03.2021 18:00


Engineering, 11.03.2021 18:00


Mathematics, 11.03.2021 18:00


Mathematics, 11.03.2021 18:00

Mathematics, 11.03.2021 18:00


English, 11.03.2021 18:00

Engineering, 11.03.2021 18:00

History, 11.03.2021 18:00

Spanish, 11.03.2021 18:00

Health, 11.03.2021 18:00

Mathematics, 11.03.2021 18:00