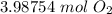CH4 + 202 → CO2 + 2H2O
How many moles of O2 needed to react with 32 grams of CH4?...

Chemistry, 30.01.2021 19:00 winstonbendariovvygn
CH4 + 202 → CO2 + 2H2O
How many moles of O2 needed to react with 32 grams of CH4?

Answers: 3
Another question on Chemistry


Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 06:00
Which change will decrease the number of effective collisions during a chemical reaction? a. adding a catalyst b. increasing the surface area c. decreasing the temperature d. increasing the reactant concentrations e. increasing the volume of the reactants
Answers: 2

Chemistry, 23.06.2019 18:40
Select the correct answer from each drop-down menu. in a reversible reaction, the forward reaction takes place the reverse reaction. such a reaction, on reaching equilibrium, will have .
Answers: 2
You know the right answer?
Questions



Mathematics, 24.04.2020 21:27


History, 24.04.2020 21:27



History, 24.04.2020 21:27






Mathematics, 24.04.2020 21:27



Mathematics, 24.04.2020 21:27


Mathematics, 24.04.2020 21:27

Business, 24.04.2020 21:27

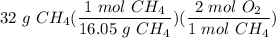 Divide/Multiply:
Divide/Multiply: