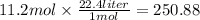50POINTS!
What is the volume of a 11.2 moles of carbon dioxide gas at STP?
A. 0.993 L<...


Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Describe in detail the melting point behavior of the 80: 20 benzoic acid-mandelic acid mixture
Answers: 3


Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
Questions




Chemistry, 24.12.2020 15:50






Computers and Technology, 24.12.2020 15:50






Biology, 24.12.2020 15:50