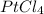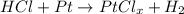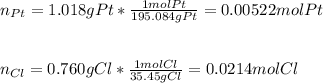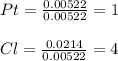Chemistry, 26.01.2021 06:10 lydia1melton
A 1.018 g sample pure platinum metal was reacted with HCl to form 1.778 g of a compound containing only platinum and chlorine. Determine the empirical formula of this "Pt-Cl" Compound.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

You know the right answer?
A 1.018 g sample pure platinum metal was reacted with HCl to form 1.778 g of a compound containing o...
Questions

English, 18.02.2021 19:30


Mathematics, 18.02.2021 19:30



Mathematics, 18.02.2021 19:30

Mathematics, 18.02.2021 19:30


History, 18.02.2021 19:30



Biology, 18.02.2021 19:30




Mathematics, 18.02.2021 19:30



Mathematics, 18.02.2021 19:30