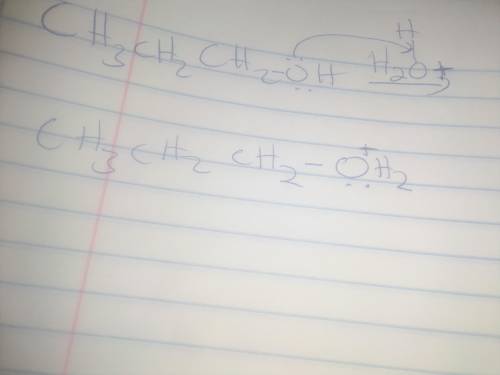When propyl alcohol is treated with acid, the initially formed intermediate is known as an oxonium ion. There is a scheme of a reversible chemical reaction. The substrates are CH3CH2CH2OH molecule and H with a charge of 1 plus ion. The product is CH3CH2CH2OH2 with a charge of 1 plus ion. Oxygen atom in CH3CH2CH2OH molecule has 2 lone pairs. Oxygen atom in CH3CH2CH2OH2 with a charge of 1 plus ion has a lone pair. All bonds are single. Using the curved arrow formalism, show how this process most likely occurs.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3


Chemistry, 22.06.2019 19:30
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1

Chemistry, 23.06.2019 00:30
What is calcium oxide+diphosphorus pentoxide--> calcium phosphate balanced
Answers: 1
You know the right answer?
When propyl alcohol is treated with acid, the initially formed intermediate is known as an oxonium i...
Questions


Geography, 09.06.2021 21:40

English, 09.06.2021 21:40


Mathematics, 09.06.2021 21:40


Chemistry, 09.06.2021 21:40





English, 09.06.2021 21:40


Arts, 09.06.2021 21:40



Mathematics, 09.06.2021 21:40

Mathematics, 09.06.2021 21:40

Physics, 09.06.2021 21:40

Biology, 09.06.2021 21:40