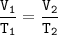Chemistry, 24.01.2021 14:00 gstinson98
A volume of 2.15 mL of Oxygen was collected in the lab at a temperature of 17 °C and 740.2 mmHg. After a day, the volume of the gas has become 2.21 mL and the barometer reading (for air pressure) had not changed. What is the new temperature in the lab (in °C)?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which is the layer underground where all empty spaces are filled with a combination of air and water ?
Answers: 1

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
A volume of 2.15 mL of Oxygen was collected in the lab at a temperature of 17 °C and 740.2 mmHg. Aft...
Questions

Mathematics, 17.12.2019 03:31

Mathematics, 17.12.2019 03:31


Mathematics, 17.12.2019 03:31


English, 17.12.2019 03:31

Mathematics, 17.12.2019 03:31

Social Studies, 17.12.2019 03:31


Mathematics, 17.12.2019 03:31






History, 17.12.2019 03:31




Mathematics, 17.12.2019 03:31