HELP ASAP! I will give brainiest if you answer all questions correctly.
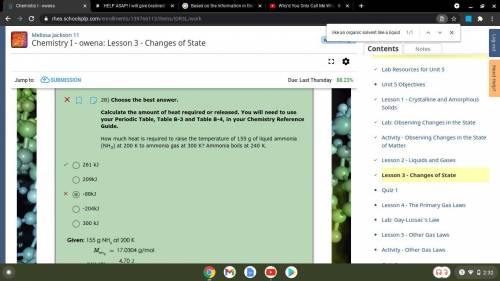
28) Choose the best answer.
Calculate the amount of heat required or released. You will need to use your Periodic Table, Table B-3 and Table B-4, in your Chemistry Reference Guide.
How much heat is required to raise the temperature of 155 g of liquid ammonia (NH3) at 200 K to ammonia gas at 300 K? Ammonia boils at 240 K.
261 kJ
-204kJ
209kJ
-88kJ
300 kJ
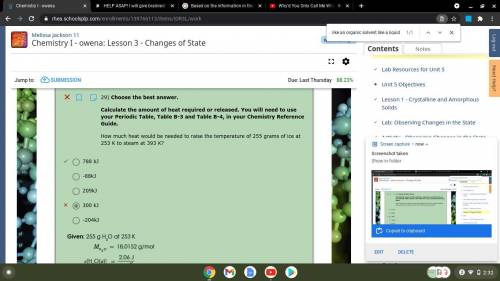
29) Choose the best answer.
Calculate the amount of heat required or released. You will need to use your Periodic Table, Table B-3 and Table B-4, in your Chemistry Reference Guide.
How much heat would be needed to raise the temperature of 255 grams of ice at 253 K to steam at 393 K?
-204kJ
300 kJ
209kJ
-88kJ
788 kJ
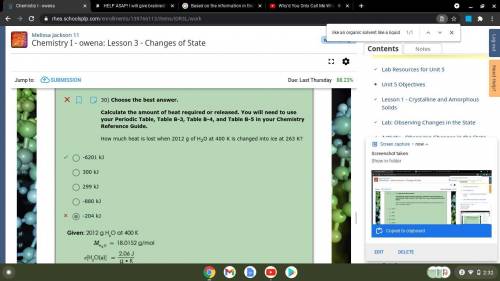
30) Choose the best answer.
Calculate the amount of heat required or released. You will need to use your Periodic Table, Table B-3, Table B-4, and Table B-5 in your Chemistry Reference Guide.
How much heat is lost when 2012 g of H2O at 400 K is changed into ice at 263 K?
300 kJ
-880 kJ
299 kJ
-204 kJ
-6201 kJ

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1

Chemistry, 23.06.2019 02:50
For questions 1 and 2, consider the following experimental data.hydrogen emission lines were detected at the following wavelengths (in nm): 121.6102.697.395.093.8question 1use the electromagnetic radiation classifications below and figure 1-1 in the introductory information for this lab (in the lab manual) to determine the nf value for the experimental data provided? wavelength, ? (nm) 650 700 550 600 400 450 500 visible spectrum wavelength, ? (m) 11 10 3 10 10 10 8 10 5 10 10 -10 10 9 10 10 10 10 -12 10 microwave radio infrared x-ray ultraviolet gamma 1020 1019 1018 1 1016 015 1014 01 12 109108 frequency, v (hz)a.1b. 2c. 3d. 4e. 5question 2using the data for the emission line with the longest wavelength, the known value of nf (from question 1 in this prelab), and the value of ni (deduced from the ? and nf values) calculate the rydberg constant for hydrogen (rh) in units of m-1.a) 1.097 x 10-11 m-1b) 5.921 x 107 m-1c) 1.097 x 10-2 m-1d) 9.252 x 106 m-1e) 1.097 x 107 m-1
Answers: 3

Chemistry, 23.06.2019 03:10
Which is true according to the law of conservation of energy
Answers: 1
You know the right answer?
HELP ASAP! I will give brainiest if you answer all questions correctly.
28) Choose the best answer....
Questions


Mathematics, 17.06.2021 19:30







Chemistry, 17.06.2021 19:30



Mathematics, 17.06.2021 19:30

English, 17.06.2021 19:30



History, 17.06.2021 19:30


Mathematics, 17.06.2021 19:30