APPLY YOUR KNOWLEDGE
Graphite and diamond are different forms of the element carbon.
Graphite...

Chemistry, 22.01.2021 14:00 anacecilianr2325
APPLY YOUR KNOWLEDGE
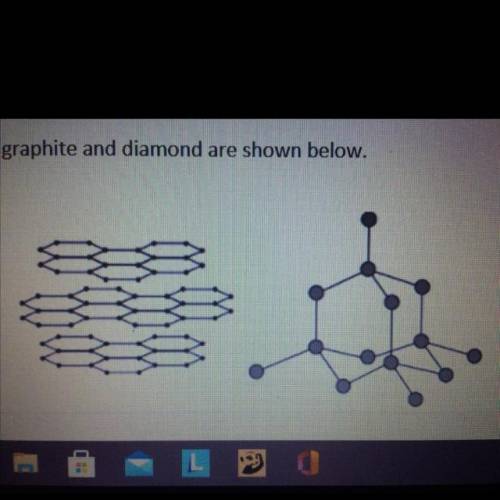
Graphite and diamond are different forms of the element carbon.
Graphite and diamond have different properties.
The structures of graphite and diamond are shown below.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2
You know the right answer?
Questions

History, 21.12.2020 18:10



Health, 21.12.2020 18:10

English, 21.12.2020 18:10





Physics, 21.12.2020 18:10

Mathematics, 21.12.2020 18:10



Mathematics, 21.12.2020 18:10



Spanish, 21.12.2020 18:10

German, 21.12.2020 18:10




