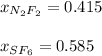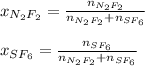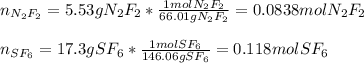Chemistry, 21.01.2021 23:00 darknessmidnight207
A 8.00 L tank at 26.9 C is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
1) this is the structure in the cell nucleus that houses a cell's genetic information
Answers: 3

Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 09:00
What factor besides temperature affects the boiling point of water? a. mass b. number of moles c. volume d. pressure
Answers: 3
You know the right answer?
A 8.00 L tank at 26.9 C is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexa...
Questions


Mathematics, 14.01.2021 17:20



English, 14.01.2021 17:20





Mathematics, 14.01.2021 17:20

Mathematics, 14.01.2021 17:20




Mathematics, 14.01.2021 17:20

Mathematics, 14.01.2021 17:20