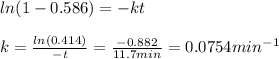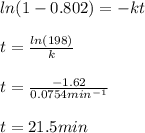Chemistry, 21.01.2021 22:20 DerekMoncoal
In a first-order decomposition reaction. 58.6% of a compound decomposes in 11.7 min. How long (in min) does it take for 80.2% of the compound to decompose

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 00:30
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1
You know the right answer?
In a first-order decomposition reaction. 58.6% of a compound decomposes in 11.7 min. How long (in mi...
Questions

Social Studies, 21.11.2020 23:30


Social Studies, 21.11.2020 23:30


Social Studies, 21.11.2020 23:30

Biology, 21.11.2020 23:30


Mathematics, 21.11.2020 23:30

Mathematics, 21.11.2020 23:30

Mathematics, 21.11.2020 23:30

Social Studies, 21.11.2020 23:30


English, 21.11.2020 23:30

Mathematics, 21.11.2020 23:30


Mathematics, 21.11.2020 23:30




![\frac{[A]}{[A]_0}=exp(-kt)](/tpl/images/1053/6847/9e1e3.png)