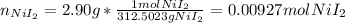Suppose 2.90g of nickel(II) iodide is dissolved in 150ml of a 0.70M aqueous solution of potassium carbonate. Calculate the final molarity of iodide anion in the solution. You can assume the volume of the solution doesn't change when the nickel(II) iodide is dissolved in it. Round your answer to 3 significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3
You know the right answer?
Suppose 2.90g of nickel(II) iodide is dissolved in 150ml of a 0.70M aqueous solution of potassium ca...
Questions



Mathematics, 28.06.2019 23:00


History, 28.06.2019 23:00

Mathematics, 28.06.2019 23:00

Social Studies, 28.06.2019 23:00


Biology, 28.06.2019 23:00

Mathematics, 28.06.2019 23:00

Mathematics, 28.06.2019 23:00



Mathematics, 28.06.2019 23:00

Computers and Technology, 28.06.2019 23:00

English, 28.06.2019 23:00



Mathematics, 28.06.2019 23:00