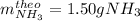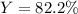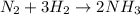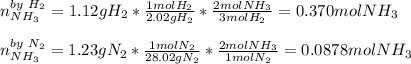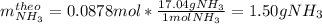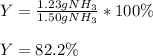Chemistry, 19.01.2021 18:50 jhernandezvaldez142
1.12g H2 is allowed to react with 9.60 g N2, producing 1.23 g NH3.
A. What is the theoretical yield in grams for this reaction under the given conditions?
B. What is the percent yield for this reaction under the given conditions?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Which best describes why nh4+ can form an ionic bond with ci-?
Answers: 1

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2
You know the right answer?
1.12g H2 is allowed to react with 9.60 g N2, producing 1.23 g NH3.
A. What is the theoretical yield...
Questions


History, 26.01.2020 23:31


History, 26.01.2020 23:31

Mathematics, 26.01.2020 23:31




Chemistry, 26.01.2020 23:31


Mathematics, 26.01.2020 23:31


Chemistry, 26.01.2020 23:31

Biology, 26.01.2020 23:31


Mathematics, 26.01.2020 23:31

Mathematics, 26.01.2020 23:31


Mathematics, 26.01.2020 23:31