
Chemistry, 19.01.2021 14:10 dragongacha777
5.Calculate the number of moles of 18.0 dm3 of carbon monoxide (CO) at rtp.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

You know the right answer?
5.Calculate the number of moles of 18.0 dm3 of carbon monoxide (CO) at rtp....
Questions



Biology, 24.07.2019 15:30




Chemistry, 24.07.2019 15:30



Computers and Technology, 24.07.2019 15:30


Mathematics, 24.07.2019 15:30

Mathematics, 24.07.2019 15:30

English, 24.07.2019 15:30





Mathematics, 24.07.2019 15:30

Social Studies, 24.07.2019 15:30

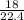 = 0.81mole
= 0.81mole 

