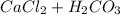Chemistry, 19.01.2021 03:20 bellbradshaw16
Aqueous solutions of Na2CO3 and Ca(NO3)2, 0.10 M each, are combined. A white precipitate is observed in the container after mixing. The precipitate is filtered and carefully rinsed with distilled water to remove other ions. A sample of the precipitate is added to 100 mL of 0.1 M NaCl. A second sample of the precipitate is then added to 100 mL of 0.1 M HCl. What would be observed in each case

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2
You know the right answer?
Aqueous solutions of Na2CO3 and Ca(NO3)2, 0.10 M each, are combined. A white precipitate is observed...
Questions



Physics, 22.04.2021 22:30

Business, 22.04.2021 22:30

Mathematics, 22.04.2021 22:30

SAT, 22.04.2021 22:30

Mathematics, 22.04.2021 22:30






Social Studies, 22.04.2021 22:30


Biology, 22.04.2021 22:30

English, 22.04.2021 22:30

Mathematics, 22.04.2021 22:30

Mathematics, 22.04.2021 22:30

Arts, 22.04.2021 22:30

Mathematics, 22.04.2021 22:30

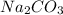 and
and 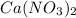 , 0.10 M each, are combined. A white precipitate is observed in the container after mixing. he precipitate is filtered andcarefully rinsed with distilled water to remove other ions. A sample of the precipitate is added to 100 mL of 0.1 M NaCl. A second sample of the precipitate is then added to 100 mL of 0.1 M HCl. What would be observed in each case?
, 0.10 M each, are combined. A white precipitate is observed in the container after mixing. he precipitate is filtered andcarefully rinsed with distilled water to remove other ions. A sample of the precipitate is added to 100 mL of 0.1 M NaCl. A second sample of the precipitate is then added to 100 mL of 0.1 M HCl. What would be observed in each case?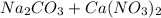 →
→ 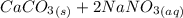
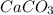 ), which, as it is insoluble in water, precipitates as a solid of the color white. This process is Precipitation and this reaction is a Precipitation Reaction.
), which, as it is insoluble in water, precipitates as a solid of the color white. This process is Precipitation and this reaction is a Precipitation Reaction. 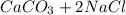 →
→ 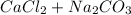
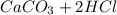 →
→