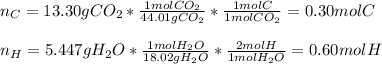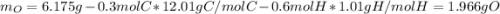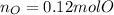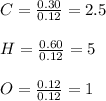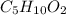Chemistry, 18.01.2021 21:40 5924000264
A 6.175 gram sample of an organic compound containing only C, H, and O is analyzed by combustion analysis and 13.30 g CO2 and 5.447 g H2O are produced. In a separate experiment, the molar mass is found to be 102.1 g/mol. Determine the empirical formula and the molecular formula of the organic compound.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1
You know the right answer?
A 6.175 gram sample of an organic compound containing only C, H, and O is analyzed by combustion ana...
Questions

Mathematics, 26.06.2019 00:00



Mathematics, 26.06.2019 00:00

History, 26.06.2019 00:00




Computers and Technology, 26.06.2019 00:00



Mathematics, 26.06.2019 00:00

Mathematics, 26.06.2019 00:00





History, 26.06.2019 00:00

Mathematics, 26.06.2019 00:00

Spanish, 26.06.2019 00:00