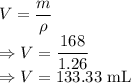Chemistry, 18.01.2021 21:40 makaylarae8781
As an instructor is preparing for an experiment, he requires 168 g glycerin. The only container readily available is a 150 mL Erlenmeyer flask. Is it large enough to contain the glycerin, whose density is 1.26 g/mL

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
As an instructor is preparing for an experiment, he requires 168 g glycerin. The only container read...
Questions

English, 14.07.2019 22:10

Social Studies, 14.07.2019 22:10

History, 14.07.2019 22:10


Mathematics, 14.07.2019 22:10


Geography, 14.07.2019 22:10

Computers and Technology, 14.07.2019 22:10


Mathematics, 14.07.2019 22:10


Biology, 14.07.2019 22:10





History, 14.07.2019 22:10

English, 14.07.2019 22:10


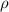 = Density of glycerin = 1.26 g/mL
= Density of glycerin = 1.26 g/mL