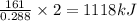Chemistry, 18.01.2021 21:30 jordandabrat
The reaction of hydrogen sulfide(g) with oxygen(g) to form water(l) and sulfur dioxide(g) proceeds as follows: 2H2S(g) 3O2(g)2H2O(l) 2SO2(g) When 9.82 g H2S(g) reacts with sufficient O2(g), 161 kJ is evolved. Calculate the value of rH for the chemical equation given. kJ/mol

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 14:30
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
The reaction of hydrogen sulfide(g) with oxygen(g) to form water(l) and sulfur dioxide(g) proceeds a...
Questions



Mathematics, 13.08.2020 20:01


Mathematics, 13.08.2020 20:01

Spanish, 13.08.2020 20:01

History, 13.08.2020 20:01

Biology, 13.08.2020 20:01


History, 13.08.2020 20:01

Mathematics, 13.08.2020 20:01

Social Studies, 13.08.2020 20:01

English, 13.08.2020 20:01

Geography, 13.08.2020 20:01


Mathematics, 13.08.2020 20:01

Medicine, 13.08.2020 20:01



Mathematics, 13.08.2020 20:01

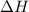 is 1118 .
is 1118 .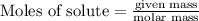
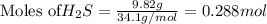
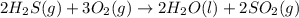
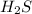 is reacted = 161 kJ
is reacted = 161 kJ